In order to improve the corrosion resistance of aluminum alloy, the anodic oxidation process of 6061 aluminum alloy with tartaric acid and sulfuric acid was optimized via orthogonal test of L16(45). Through range analysis and variance analysis, combined with oxidation film surface morphology observation, thickness test and electrochemical impedance analysis, the optimal process parameters of corrosion resistance were obtained. On the basis of the above, the effect of hole sealing on corrosion resistance was further studied. Results showed that with taking corrosion resistance as the index, the optimal process parameters were oxidation temperature of 20~25 ℃, oxidation time of 50 min, oxidation voltage of 20 V, tartaric acid concentration of 80 g/L. The oxidation film prepared under the optimal parameters possessed the thickness of 10 μm and the impedance value of 4.148×107 Ω·cm2. After sealing the hole of oxide film with boiling water, the impedance value of the intermediate frequency place of the oxidation film was improved from 1 × (103~104) Ω·cm2 to 1 × (104~105) Ω·cm2. From the experiments, it could be found that there was no correlation between oxidation film thickness and corrosion resistance, and the key factors affecting corrosion resistance is oxidation time and tartaric acid concentration. Besides, Boiling water sealing treatment effectively improved the corrosion resistance of the oxide film.


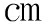 2提高到1×(104~105) Ω·
2提高到1×(104~105) Ω·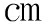 2左右。试验发现氧化膜厚度与耐蚀性之间没有相关性,影响耐蚀性的关键因素是氧化时间和酒石酸浓度。沸水封孔处理有效提高了氧化膜的耐蚀性。
2左右。试验发现氧化膜厚度与耐蚀性之间没有相关性,影响耐蚀性的关键因素是氧化时间和酒石酸浓度。沸水封孔处理有效提高了氧化膜的耐蚀性。